|
These findings are considered a class effect of LABA monotherapy. Available data from controlled clinical trials also suggest that use of LABA as monotherapy increases the risk of asthma-related hospitalization in pediatric and adolescent patients. Use of long-acting beta 2-adrenergic agonists (LABA) as monotherapy (without ICS) for asthma is associated with an increased risk of asthma-related death. Warnings and Precautions Serious Asthma-Related Events – Hospitalizations, Intubations, Death Severe hypersensitivity to milk proteins or demonstrated hypersensitivity to fluticasone furoate, umeclidinium, vilanterol, or any of the excipients.Primary treatment of status asthmaticus or other acute episodes of COPD or asthma where intensive measures are required.Trelegy Ellipta is contraindicated in the following conditions: 
200 mcg fluticasone furoate, 62.5 mcg umeclidinium, and 25 mcg vilanterol (200/62.5/25 mcg) per actuation.100 mcg fluticasone furoate, 62.5 mcg umeclidinium, and 25 mcg vilanterol (100/62.5/25 mcg) per actuation.If asthma symptoms arise in the period between doses, an inhaled, short-acting beta2-agonist (rescue medicine, e.g., albuterol) should be used for immediate relief.For patients who do not respond adequately to Trelegy Ellipta 200/62.5/25 mcg once daily, re-evaluate and consider other therapeutic regimens and additional therapeutic options. For patients who do not respond adequately to Trelegy Ellipta 100/62.5/25 mcg once daily, increasing the dose to Trelegy Ellipta 200/62.5/25 mcg once daily may provide additional improvement in asthma control.The maximum recommended dosage is 1 inhalation of Trelegy Ellipta 200/62.5/25 mcg once daily.When choosing the starting dosage strength of Trelegy Ellipta, consider the patients’ disease severity their previous asthma therapy, including the inhaled corticosteroid (ICS) dosage as well as the patients’ current control of asthma symptoms and risk of future exacerbation.The recommended starting dosage of Trelegy Ellipta for maintenance treatment of asthma is fluticasone furoate 100 mcg, umeclidinium 62.5 mcg, and vilanterol 25 mcg (1 actuation of Trelegy Ellipta 100/62.5/25 mcg) or fluticasone furoate 200 mcg, umeclidinium 62.5 mcg, and vilanterol 25 mcg (1 actuation of Trelegy Ellipta 200/62.5/25 mcg) once daily, by oral inhalation. Recommended Dosage for Maintenance Treatment of Asthma If shortness of breath occurs in the period between doses, an inhaled, short-acting beta 2-agonist (rescue medicine, e.g., albuterol) should be used for immediate relief.Trelegy Ellipta 100/62.5/25 mcg is the only strength indicated for the treatment of COPD.The recommended dosage of Trelegy Ellipta for maintenance treatment of COPD is fluticasone furoate 100 mcg, umeclidinium 62.5 mcg, and vilanterol 25 mcg (1 actuation of Trelegy Ellipta 100/62.5/25 mcg) once daily by oral inhalation. Recommended Dosage for Maintenance Treatment of Chronic Obstructive Pulmonary Disease No dosage adjustment is required for geriatric patients, patients with renal impairment, or patients with moderate hepatic impairment.Do not use Trelegy Ellipta more than 1 time every 24 hours. Trelegy Ellipta should be used at the same time every day.After inhalation, rinse the mouth with water without swallowing to help reduce the risk of oropharyngeal candidiasis.Administer 1 actuation of Trelegy Ellipta once daily by oral inhalation.Trelegy Ellipta Dosage and Administration Dosage and Administration Overview Trelegy Ellipta is NOT indicated for the relief of acute bronchospasm. Trelegy Ellipta is indicated for the maintenance treatment of asthma in patients aged 18 years and older. Trelegy Ellipta is indicated for the maintenance treatment of patients with chronic obstructive pulmonary disease (COPD). 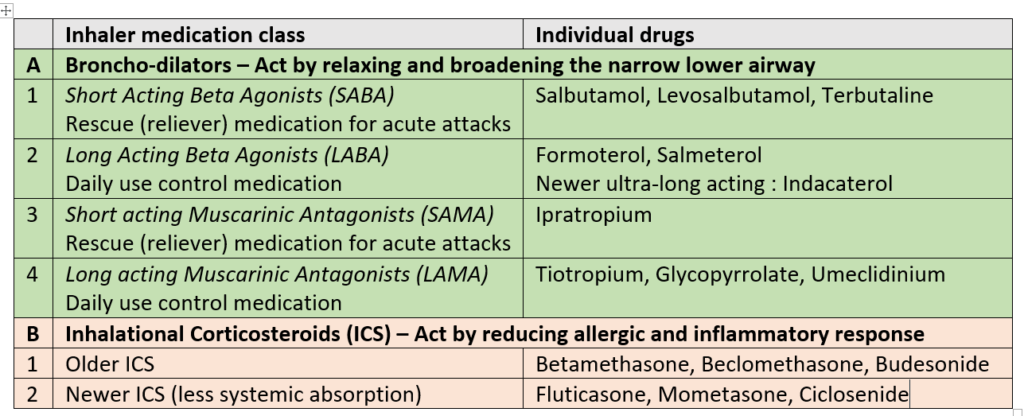
Expand Indications and Usage for Trelegy Ellipta Maintenance Treatment of Chronic Obstructive Pulmonary Disease
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
